An international team of scientists has developed a cheap way to provide fresh water to thirsty communities by making seawater drinkable without using electricity.
So long as the sun is shining, they say, their device will produce enough high-quality potable water to cover a family’s needs, at a cost of around $100.
The scientists, from Massachusetts institute of Technology (MIT), U.S. and Shanghai Jiao Tong University, China, believe their brainwave offers a simple solution to thirsty islands and arid coastal areas which lack a reliable electricity supply but have access to seawater. It could even help to prevent some of the mass migrations expected with climate change.
The researchers report their work in the journal Energy and Environmental Science. Testing their prototype on a roof at the Massachusetts Institute of Technology, they produced more than 1.5 gallons of fresh drinking water every hour for every square meter of solar collecting area.
Their device is cube-shaped, with multiple layers of solar evaporators and condensers piled one on top of another, surmounted with a layer of transparent insulation. Essentially it is a multi-layer solar still, similar to those used for centuries to make strong liquor and used today in many applications.
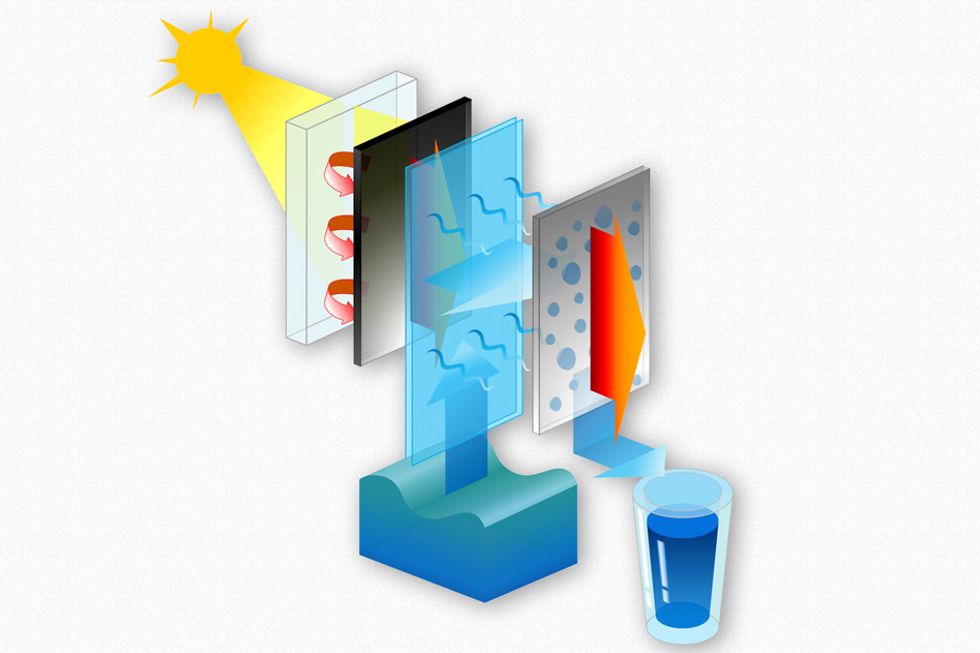
Diagram illustrates the basic structure of the proposed desalination system. Sunlight passes through a transparent insulating layer at left, to heat up a black heat-absorbing material, which transfers the heat to a layer of wicking material (shown in blue), where it evaporates and then condenses on a surface (gray) and then drips off to be collected as fresh, potable water. Images courtesy of the researchers
A solar still uses flat panels to absorb heat which it then transfers to a layer of water, which begins to evaporate. The vapor condenses on the next panel and the water is collected, while the heat from the vapor condensation is passed to the layer above.
Whenever vapor condenses on a surface, it releases heat; in typical condenser systems, that heat is simply lost to the environment. But in this multi-layer version the released heat flows to the next evaporating layer, recycling the solar heat and boosting overall efficiency.
The efficiency comes from using each of the multiple stages to remove salt from the sea water, with the heat released by the previous stage harnessed instead of wasted. In this way, the team’s demonstration device achieved an overall efficiency of 385 percent in converting the energy of sunlight into evaporation.
Evelyn Wang, a co-author, said: “When you condense water, you release energy as heat. If you have more than one stage, you can take advantage of that heat.”
Cost Trade-Off
Although adding more layers increases the conversion efficiency of the system, each layer also adds cost and bulk. The team settled on a 10-stage system for their proof-of-concept device.
It delivered pure water that exceeded city drinking water standards, at a rate of 5.78 liters per square meter (about 1.52 gallons per 11 square feet) of solar collecting area. This is more than twice as much as the record amount previously produced by any such passive solar-powered desalination system, Professor Wang says.
And a big advantage of the system is that it has a self-flushing mechanism which will clean out the accumulation of salt each night and return it to the sea.
One possible way of using the system would be with floating panels on a body of saltwater. The panels could deliver constant fresh water through pipes to the shore so long as the sun was shining. Other systems could be designed to serve a single household, perhaps using a flat panel on a large shallow tank of seawater.
The team estimates that a system with a roughly one-square-meter solar collecting area could meet the daily drinking water needs of one person. In production, they think a system built to serve the needs of a family might be built for around $100.
Cheaper Replacements
The most expensive component of the prototype is the layer of transparent aerogel used as an insulator at the top of the stack, but the team suggests other less expensive insulators could be used instead. (The aerogel itself is made from very cheap silica but requires specialized drying equipment during its manufacture.)
“This new approach is very significant,” says Professor Ravi Prasher of Lawrence Berkeley National Laboratory and the University of California at Berkeley, who was not involved in the research.
“One of the challenges in solar still-based desalination has been low efficiency due to the loss of significant energy in condensation.
“By efficiently harvesting the condensation energy, the overall solar to vapour efficiency is dramatically improved … This increased efficiency will have an overall impact on reducing the cost of produced water.”